|
6/19/2023 0 Comments Entropy table 
Steam Consumption for some Typical Steam Heated Consumers Boiler PressureĬalculate the stress in steam boiler shells caused by steam pressure. Steam consumption and condensate generation when heating liquid or gas flows Steam - Flow vs. Steam is a compressible gas where pipe line mass flow capacity depends on steam pressure. 
Steam table with sensible, latent and total heat, and specific volume at different gauge pressures and temperatures. Saturated Steam - Properties with Imperial Units Saturated Steam Table with properties like boiling point, specific volume, density, specific enthalpy, specific heat and latent heat of vaporization. When flash steam is generated and vented to the surroundings a considerable amount of energy is lost Saturated Steam - Properties for Pressure in Bar Evaporative CoolingĮvaporative cooling tutorial. Evaporation from a Water SurfaceĮvaporation of water from a water surface - like a swimming pool or an open tank - depends on water temperature, air temperature, air humidity and air velocity above the water surface - online calculator. Condensate Pipes - Flash Steam GeneratedĬalculate flash steam generation in condensate pipe lines. Specific volume, enthalpy and entropy of compressed water. Related Documents Arithmetic and Logarithmic Mean Temperature DifferenceĪrithmetic Mean Temperature Difference in Heat Exchangers - AMTD - and Logarithmic Mean Temperature Difference - LMTD - formulas with examples - Online Mean Temperature Calculator. Thermodynamics of steam and condensate systems. Steam & condensate systems- properties, capacities, pipe sizing, systems configuration and more. In other words the same pipe may transfer more energy with high pressure steam than with low pressure steam.įlash steam generation - thermodynamic fundamentals, heat loss, energy recovery and more. Note! Because the specific volume of steam decreases with increasing pressure, the amount of heat energy transferred in the same volume actually increases with steam pressure. The evaporation heat is 2047 kJ/kg according the table. The specific enthalpy of evaporation decrease with steam pressure increase. 
The heat energy (enthalpy of evaporation) needed at 7 bar gauge to evaporate the water to steam is actually less than the heat energy required at atmospheric pressure. From the table a value of 720.9 kJ is needed to raise 1 kg of water from 0 oC to the saturation temperature 170 oC. More heat energy is required to raise the temperature to the saturation point at 7 bar gauge than needed for water at atmospheric pressure. Steam at atmospheric pressure is of limited practical use since it can not be conveyed by its own pressure along a steam pipe to the points of consumption.Īt 7 bar gauge (absolute 800 kN/m 2) - the saturation temperature of water is 170 oC. (1 hour = 3600 seconds, 1 kW = 1 kJ/s) Example - Boiling Water at 170 oC and 7 bar The total specific enthalpy of the steam (or heat required to evaporate water to steam) at atmospheric pressure and 100 oC can be summarized as: Therefore, at 0 bar gauge (absolute 101.33 kN/m 2) - the specific enthalpy of evaporation is 2257 kJ/kg. Therefore, at 0 bar gauge (absolute 101.33 kN/m 2) and 100 oC - the specific enthalpy of water is 419 kJ/kg.Īnother 2257 kJ of energy is required to evaporate the 1 kg of water at 100 oC to steam at 100 oC. 419 kJ of energy is required to heat 1 kg of water from 0 oC to the saturation temperature 100 oC. Specific enthalpy - or Sensible Heat - is the quantity of heat in 1 kg of water according to the selected temperatureĮxample - Boiling Water at 100 oC and 0 barĪt atmospheric pressure - 0 bar gauge or absolute 101.33 kN/m 2- water boils at 100 oC.Absolute Pressure = Gauge Pressure + Atmospheric Pressure. 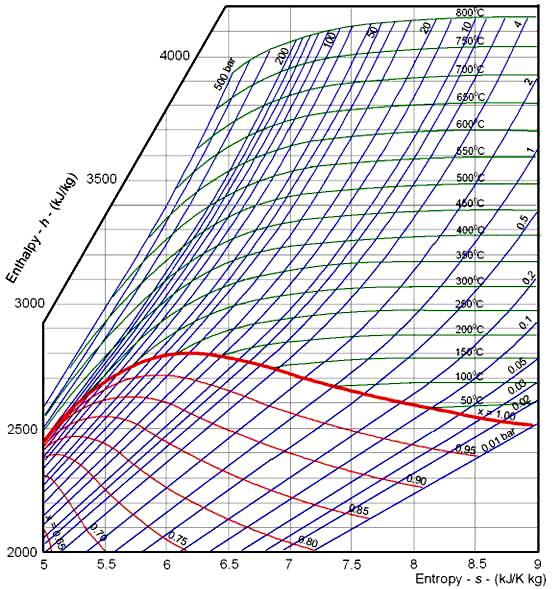
Vacuum steam is the general term used for saturated steam at temperatures below 100☌.Therefore, the distance traveled is not a state function and is not pathway independent. If you go straight up, the distance will differ from if you zigzag and for any other path you take. On the other hand, the distance you travel up the mountain does depend on the path you take. This makes this function pathway independent. Whether you went straight up, zigzagged, went curvily, or any other pathway, your change in altitude will be the same at the end. Since no matter which way you go, you will end up at the top of the mountain, your change in altitude will always be the altitude. An example of a state function is the change in altitude when climbing a mountain, whereas an example of a non-state function is the distance traveled. This means that whatever “path” you take to get to the end result, the end result will be exactly the same. In essence, a state function is a function that has the property of pathway independence. Let’s take a quick side note to explain what a state function means and why it’s important. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
